Introduction
DISC-0974 is a monoclonal antibody (mAB) targeting hemojuvelin and is currently in clinical trials evaluating effects on anemia in patients with myelofibrosis or chronic kidney disease. Inhibition of hemojuvelin (HJV) results in decreased hepcidin and reduction in ferroportin degradation, enabling more iron to be exported out of cells and into the bloodstream for transport to other cells and tissues.
Methods
A study was conducted in healthy, naïve cynomolgus monkeys to evaluate the safety, pharmacokinetics (PK), and pharmacodynamics (PD) of DISC-0974 following once-monthly dosing for 6 months. Male and female cynomolgus monkeys (n=6/sex/dose group) were intravenously administered DISC-0974 at doses of 0, 0.3, 1, or 6 mg/kg on Days 1, 29, 57, 85, 113, and 141. An additional IV dose was administered on Day 165 for males and Day 166 for females. Dose levels were selected where the high dose would maximally mobilize iron for sustained periods over the treatment regimen, with lower doses selected to provide a dose-response. Male and female monkeys (n=4/sex/group) were euthanized on Day 168 and 169, respectively, and subgroups were monitored for an additional 6 months (n=2/sex/group) to evaluate reversibility of any effects. PK was evaluated after the 1 st, 3 rd, and 6 th dose in all monkeys, as well as during the 6-month period after the final dose in the remaining monkeys. PD effects (i.e., serum iron and transferrin saturation [TSAT]) were monitored at the same timepoints as those collected for PK.
Results
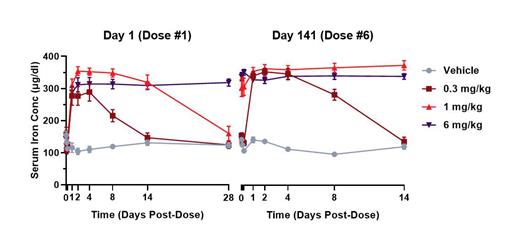
DISC-0974 was well tolerated, and no DISC-0974-related adverse effects were observed. Systemic exposure, based on area under the curve (AUC) and maximum plasma concentrations (C max), increased with dose. Systemic exposures were achieved that reached and exceeded those observed in clinical trials. The high dose of 6 mg/kg achieved systemic exposures, based on AUC, that were >15-fold those observed in humans. Half-lives were consistent with an mAb, and repeated exposure did not result in accumulation with monthly dosing. Increases in serum iron (up to approximately 3-fold baseline) (Figure 1) and TSAT (>90%), representative of PD activity, correlated with systemic exposures of DISC-0974, with similar activity maintained after each of the 7 monthly doses.
Conclusion
Overall, these data demonstrate that DISC-0974 is well tolerated after repeated monthly exposure in cynomolgus monkeys, and an appropriate and durable PK/PD profile was observed, supporting clinical development of chronic once-monthly dosing.
Disclosures
Reisman:Disc Medicine: Current Employment, Current equity holder in publicly-traded company. Savage:Disc Medicine: Current Employment, Current equity holder in publicly-traded company, Divested equity in a private or publicly-traded company in the past 24 months. Liu:Disc Medicine: Current Employment, Current equity holder in publicly-traded company. Yang:Disc Medicine: Current Employment, Current equity holder in publicly-traded company.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal